 It is the basic unit of all light carrying the energy E= hf.Ītomic emission spectrum: is the pattern of lines corresponds to a different electron transition from a higher energy state to a lower energy state. Photon: the smallest discrete amount of electromagnetic radiation. Every element has a unique atomic absorption and emission line spectrum.Įxcited-state: of an atom is a state where its potential energy is higher than the ground state.The electromagnetic radiation in both cases has some type of energy with a specific wavelength. The atomic emission spectrum is the pattern of lines formed when light passes through a prism to separate it into the different frequencies of light it contains. Emission is when a substance gives off electromagnetic radiation. 
In the second example below, where an element has 3 excited states, it could emit photons at 6 specific wavelengths/frequencies (ΔE= hf). Every element has a unique atomic absorption and emission line spectrum. An atomic emission spectrum is the pattern of lines formed when light passes through a prism to separate it into the different frequencies of light it contains.Įach of these spectral lines corresponds to a different electron transition from a higher energy state to a lower energy state. When a narrow beam of this light was viewed through a prism, the light was separated into four lines of particular wavelengths. Scientists studied the distinctive pink color of the gas discharge created by hydrogen gas. Signs of other colors contain different gases or mixtures of gases. 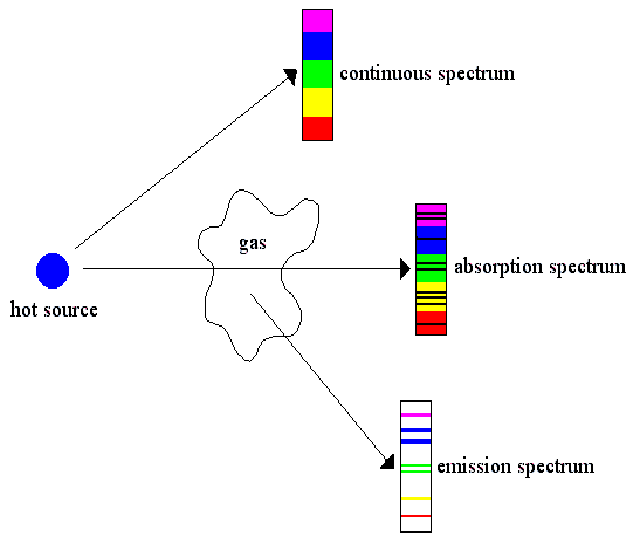
(b) Discus the similarities and differences between emission spectra and absorption. However, only signs that glow with the red-orange color seen in the figure are filled with neon. (a) Decribe the appearance and origin of an emission line spectrum. What are Emission Spectra Definition, Characteristics 3. What are Absorption Spectra Definition, Characteristics 2. “Neon” signs are familiar examples of gas discharge tubes. The main difference between absorption and emission spectra is that absorption spectra show black colored gaps/lines whereas emission spectra show different colored lines in the spectra. 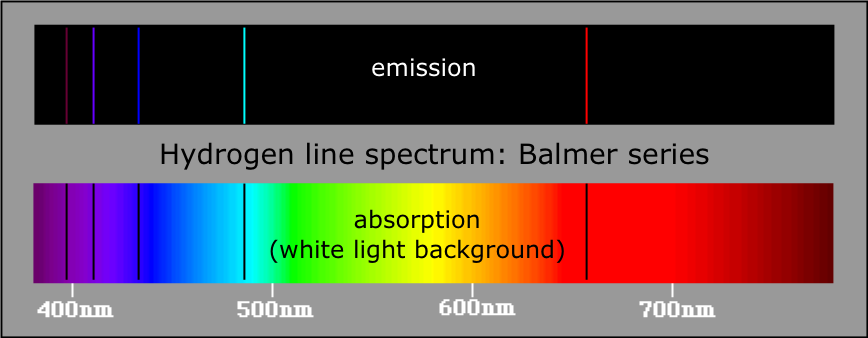
Electrons in the gaseous atoms first become excited, and then fall back to lower energy levels, emitting light of a distinctive color in the process. These gas discharge tubes are enclosed glass tubes filled with a gas at low pressure through which an electric current is passed. Since the electron energy levels are unique for each element, every gas discharge tube will glow with a distinctive color depending on the identity of the gas. One way for atoms to gain energy is to pass an electric current through an enclosed sample of a gas at low pressure called a gas discharge tube. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
